Important Results
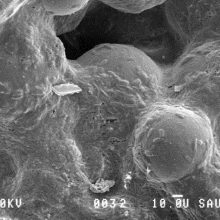
1) The manipulative experiments conducted in the laboratory showed that biofilm growth reduced sediment erodibility, leading to an average 15-fold increase in sediment stability.
2) The stabilizing effect of biofilms varied in magnitude depending on the external boundary conditions (an increase in sediment stability was observed for higher bed shear stresses and smaller mean grain sizes for the GBS).
3) Positive correlations exist between extracellular polymeric substances (EPS), biomass and sediment stability, indicating a sediment binding potential of the EPS matrix.
4) However, the increased sediment stability cannot be attributed to biochemical sediment properties alone, but also to binding by filamentous bacteria.
Brief summary
Motivation: Exploring biological mediation of sediments – its role and significance in reservoirs
Novelty: Linking biostabilisation to seasonal and operationally-based pattern & algal blooms
Strategy: Field samples and manipulative experiments in the laboratory
Methods: Magnetic Particle Induction MagPI (Biofilm-Adhesion), Floc Erosion Chamber and Floc Settling Column, Camera plus evaluation software (Floc characteristics), Chemistry (Extracellular Polymeric Substances EPSs), Microbiology (Cell numbers, Biomass, Microbial Community)
Expected results: Better prediction of siltation and water quality at different management modes

(A) Motivation: The dynamic of fine cohesive sediments is currently unpredictable and poorly understood despite its vital role in reservoirs. There is a consensus that organic matter and biological activities significantly affect the behaviour of fine sediments, but interlinks between triggers (e.g. environmental parameters) and processes (e.g. EPSs secretion) are still to be unravelled in limnic habitats. Yet, to fully understand the role and binding capacity of biostabilisation for sediment stability and consolidation as well as flocculation behaviour is the prerequisite for improved management strategies in reservoirs.
(B) Aims: Consequently, the overall objective of WP2 is to examine for the first time the significance of biostabilisation within reservoirs and the extent to which it is important for the deposition and remobilisation of fine sediments. In this context, possible variations in biostabilisation due to seasonal changes and specific environmental as well as biotic conditions at various locations have to be addressed while following these main research questions: (1) Can we see clear seasonal, biotic (=e.g. grazing) and operationally-based (abiotic) pattern in biostabilisation within reservoirs that are reflected in e.g. biofilm topography/architecture, community, metabolic activity and functionality (together with WP3)? (2) What is the importance of cohesive (inter-particles forces) versus adhesive (biologically-induced forces) binding for the overall stability within surface layers as well as within deeper sediment horizons (together with WP1)? (2) Do cyanobacteria contribute to stabilisation and do biofilms serve as reservoirs of toxic algal blooms (together with WP3)?
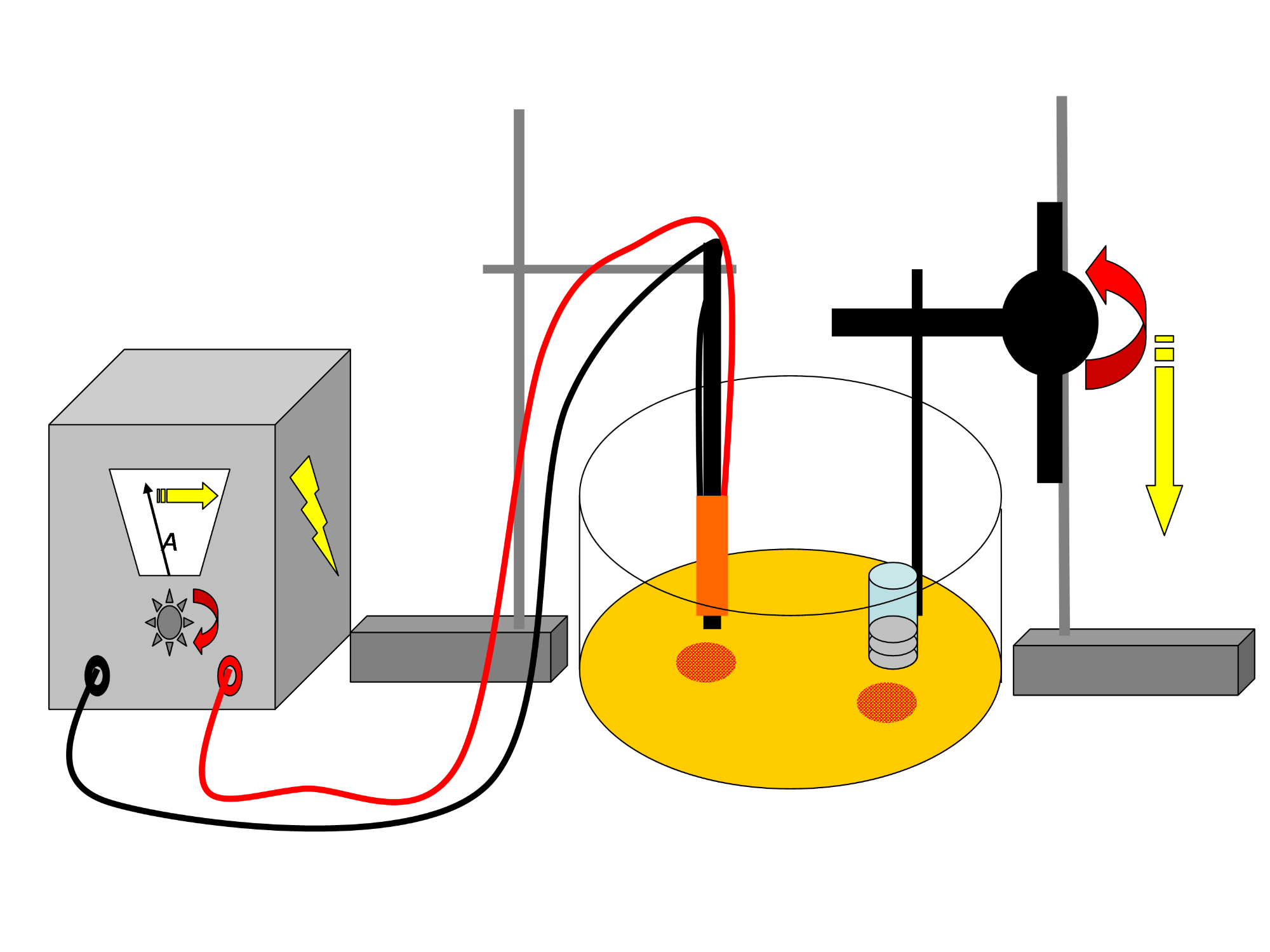
(C) Methods: In general, the project will concern the performance of manipulative experiments in the laboratory to gain a deeper process understanding plus the analysis of field samples. WP2 is responsible (together with WP3) to describe the microbial community at various reservoirs. WP2 will determine microalgae biomass, bacterial cell numbers as well as the microalgae species community composition by microscopy (diatoms, cyanobacteria, and chlorophytes) while WP3 will perform the molecular analyses. These microbial parameters will be linked to sediment characteristics such as particles sizes, TOC, and CEC determined by WP1. Moreover, WP2 will complement WP1 measurements of sediment stability and resuspended material by determination of the adhesive capacity (applying Magnetic Particle Induction/MagPI) and floc characteristics (using a floc erosion and settling chamber, camera and evaluation software), respectively.
In manipulative laboratory experiments (with WP3), water of the particular reservoir will serve as an inoculum for microbes forming natural biofilms on artificial substrates. In our mesocosm (DFG project GZ: GE 1932/3-2, Schmidt et al. 2015), these biofilms will be exposed to various scenarios of hydrodynamics (up to desiccation), seasons and biotic influence (such as grazing) to unravel links between biofilm topography, community composition, physiology and functionality (here biostabilisation). In all of the planned experiments, the growth of the biofilm will be regularly monitored by determining microbial (biomass, cell numbers, community composition) and chemical parameters (extracellular polymeric substances/EPS, here sugars and proteins) and by measuring the biostabilisation capacity (MagPI, SETEG) and flocculation (floc erosion chamber, floc settling column).
(D) Achievable Goals: The species composition, the architecture and the metabolic activity of biofilms strongly depend on abiotic conditions, such as light and nutrient availability as well as hydrodynamics. Consequently, these abiotic conditions will also affect EPS secretion and thus, biostabilisation capacity and, after resuspension, flocculation. Hence, we will gain crucial knowledge on the response of biofilms to certain environmental conditions in order to better predict cohesive sediment dynamics within reservoirs. Thereby, we aim to contrast microbial communities from different seasons and locations as well as varying abiotic conditions within the reservoir and different catchments (spatial and temporal scales). Beyond the significant progress that is to be expected in the research on biofilm and biostabilisation within freshwaters, there will insights on the potential effects of different operational modes within the reservoir (changes in quantity and quality of the inflow, low and high reservoir level etc.) that will help to reveal future management options (as already pointed out above in WP1). In particular, this project will explore the linkage between microbial community composition (WP2, WP3), grazers (WP3), metabolic reactions (EPS secretion WP2, toxin production WP3), stabilisation and flocculation (WP1, WP2); all of which concern potential conflict topics such as water quality and siltation (WP5).